Adsorption
and Absorption
Absorption is the process in which a fluid is
dissolved by a liquid or a solid (absorbent).
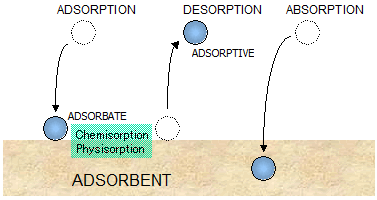
Adsorption is the process in which atoms,
ions or molecules from a substance (it could be gas, liquid or dissolved solid)
adhere to a surface of the adsorbent. Adsorption is a surface-based process
where a film of adsorbate is created on the surface while absorption involves
the entire volume of the absorbing substance.
Comparison Chart
Items
|
Absorption
|
Adsorption
|
Definition
|
Assimilation of molecular species
throughout the bulk of the solid or liquid is termed as absorption.
|
Accumulation of the molecular
species at the surface rather than in the bulk of the solid or liquid is
termed as adsorption.
|
Phenomenon
|
It is a bulk phenomena
|
It is a surface phenomena.
|
Heat Exchange
|
Endothermic process
|
Exothermic process
|
Temperature
|
It is not affected by temperature
|
It is favored by low temperature
|
Rate of Reaction
|
It occurs at a uniform rate.
|
It steadily increases and reach
to equilibrium
|
Concentration
|
It is same throughout the material.
|
Concentration on the surface of adsorbent is different
from that in the bulk
|
Process
Gas-liquid
absorption (a) and liquid-solid adsorption (b) mechanis.
Blue spheres are
solute molecules
Adsorption and
absorption are both sorption processes.
Absorption occurs when atoms pass through or enter a bulky material. During
absorption, the molecules are entirely dissolved or diffused in the absorbent
to form a solution. Once dissolved, the molecules cannot be separated easily
from the absorbent.
Adsorption is generally
classified into physisorption (weak van der Waal’s forces) and chemisorption.
It may also occur due to electrostatic attraction. The molecules are held
loosely on the surface of the adsorbent and can be easily removed.
Adsorption: Some of the
industrial applications for adsorption are air-conditioning, adsorption
chillers, synthetic resin and water purification. An adsorption chiller does
not require moving parts and hence is quiet. In pharmaceutical industry
applications, adsorption is used as a means to prolong neurological exposure to
specific drugs or parts thereof. Adsorption of molecules onto polymer surfaces
is used in various applications such as in the development of non-stick
coatings and in various biomedical devices.
Absorption: The common
commercial uses of absorption cycle are absorption chillers for space cooling
applications, ice production, cold storage, turbine inlet cooling.
High efficiency operation, environmentally friendly refrigerants,
clean-burning fuels and few moving parts that require maintenance make
absorption a very good choice for consumers.
The process of gas absorption by a liquid
is used in hydrogenation of oils and carbonation of beverages.
Gold APP
Instruments Corporation China
Mobile:
+86-18210009838